|
3/1/2023 0 Comments Ctla4 tcel 
All patients’ samples were obtained with informed consent in Samsung Medical Center, Seoul, Korea, and the study was approved by the Institutional Review Board in accordance with the Declaration of Helsinki. The QIAamp DNA Mini Kit (Qiagen) and RNeasy Mini Kit (Qiagen) were used for DNA and RNA extraction, respectively. Formalin-fixed, paraffin-embedded tumor tissue was used for Sanger sequencing of 115 TCL tumor samples and targeted deep sequencing of 74 TCL tumor samples. The patients’ clinical information is summarized in Online Supplementary Table S2. The mutational status of 70 lymphoma-associated genes was analyzed using ultra-deep targeted resequencing in 74 samples of mature TCL. A functional study of the fusion gene indicated that fusion between CTLA4 and CD28 results in the activation of downstream oncogenic pathways. 10 In this study, we identified a fusion between CTLA4 and CD28 in a case of angioimmunoblastic TCL by whole transcriptome sequencing and analyzed the frequency of gene fusion in 117 cases of TCL. 6 Accordingly, controlling T-cell signaling through these two co-receptors has been a key strategy for recent cancer immunotherapies including anti-CTLA4 antibody therapy 9 8 and chimeric antigen receptor T-cell therapy utilizing the intracellular signaling domain of CD28. 7 6 Perturbing this balance can result in a number of undesirable consequences such as autoimmunity, transplant rejection, or even malignant TCL. 
They play critical and opposite roles in maintaining balanced T-cell signaling and, thus, the proper level of immune activation. It is, therefore, still necessary to identify additional driver mutations and dissect the interplay with T-cell signaling components.ĬTLA4 and CD28, members of the immunoglobulin superfamily that are expressed on the surfaces of T cells, are regulatory co-receptors of T-cell signaling. Furthermore, none of the mutant genes has been clearly demonstrated to be a dominant oncogenic driver. 5 1 However, the roles of these mutations in the regulation of T-cell signaling and oncogenesis have yet to be elucidated. Recent genomic studies have identified highly recurrent somatic mutations in TET2, DNMT3A, IDH2, and RHOA in diverse subtypes of mature T-cell lymphoma (TCL). Peripheral T-cell lymphoma is a malignant neoplasm of mature T cells. The CTLA4-CD28 gene fusion is likely a major contributor to the pathogenesis of T-cell lymphomas and represents a potential target for anti-CTLA4 cancer immunotherapy. The mutational landscape we obtained suggests that T-cell lymphoma results from diverse combinations of multiple gene mutations. We further investigated the mutation status of 70 lymphoma-associated genes using ultra-deep targeted resequencing for 74 mature T-cell lymphoma samples. We identified the fusion in 26 of 45 cases of angioimmunoblastic T-cell lymphomas (58%), nine of 39 peripheral T-cell lymphomas, not otherwise specified (23%), and nine of 31 extranodal NK/T cell lymphomas (29%). To estimate the frequency of this gene fusion in mature T-cell lymphomas, we examined 115 T-cell lymphoma samples of diverse subtypes using reverse transcriptase polymerase chain reaction analysis and Sanger sequencing. Ectopic expression of the fusion transcript in Jurkat and H9 cells resulted in enhanced proliferation and AKT and ERK phosphorylation, indicating activation of downstream oncogenic pathways. The fusion gene, which codes for the extracellular domain of CTLA4 and the cytoplasmic region of CD28, is likely capable of transforming inhibitory signals into stimulatory signals for T-cell activation. By RNA sequencing, we identified a fusion between the two genes from partial gene duplication in a case of angioimmunoblastic T-cell lymphoma. 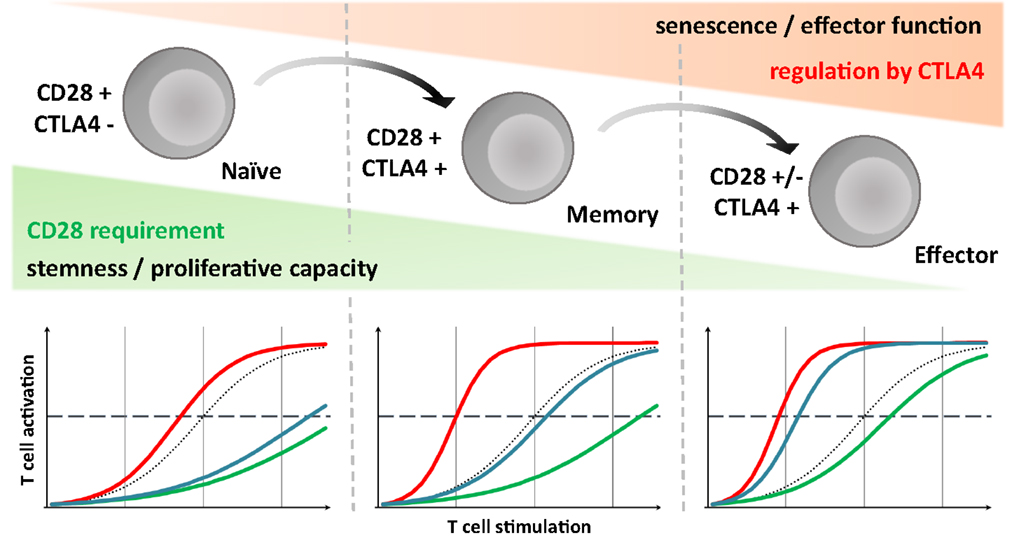
CTLA4 and CD28 are co-regulatory receptors with opposite roles in T-cell signaling. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
